|
A nonreactive (inert) platinum wire conducts electrons into the right beaker.\): A galvanic cell based on the spontaneous reaction between copper and silver(I) ions.Ībbreviated symbolism is commonly used to represent a galvanic cell by providing essential information on its composition and structure. Spectator ions are not included in the notation. The concentrations of important species in each of the half reactions are included. The notation shows the anode on the left and cathode on the right. There is a shorthand notation used to specify the conditions of an electrochemical cell. A) 2 Ag) Ag() POCI,) ARCH incare Consider these two entries from a fictional table of. Shorthand Notation for an Electrochemical Cell. Anode half-reaction: P(0) +261 POCI,(6) Cathode half-reaction: + 2e 2 ACH +2e' Agco + 2C1 Shorthand notation: Pb(s) POCI,) C(a) Cl(aq) ACI) Ag(s) Answer Bank CI' Po(s) 2. The solid, liquid, or aqueous phases within a half-cell are separated by a single line. The electrode on the left is the anode, and the one on the right is the cathode. The essential components of each half reaction are described and each piece of information is separated by a single bar (), which also represents a phase boundary. The double bars () represent the salt bridge. The salt bridge is represented by a double line. Shorthand notation used to describe electrochemical cells: anode reactioncathode reaction. Divide 300,000 by 6,000 to obtain a value of 50. Let’s plug in 300,000 J for G o to match R. Even though G o is normally expressed as kJ/mol, R is expressed as J/molK, so we can convert R or G o to match units. The oxidation of magnesium to magnesium ions occurs in the beaker on the left side in this apparatus the reduction of hydrogen ions to hydrogen occurs in the beaker on the right. In this notation, information about the reaction at the anode appears on the left and information about the reaction at the cathode on the right. We can plug in the value of G o on the left side of the equation. Question: For the galvanic cell reaction, expressed below using shorthand notation, what half-reaction occurs at the cathode Ba (s) Ba2+ (aq) Cd2+ (aq) Cd (s) A. An example of such a voltaic cell is shown below. This problem has been solved Youll get a detailed solution from a subject matter expert that helps you learn core concepts. For such substances, an inert electrode that does not participate in the reactions is used. In the reaction above, the anode is the Cu(s) since it increases in oxidation state from 0. In other words, this is where the metal loses electrons. Anode: The anode is where the oxidation reaction takes place. Some redox reactions involve species that are poor conductors of electricity, such as gases or ionic solids. When an electrode is oxidized in a solution, it is called an anode and when an electrode is reduced in solution. 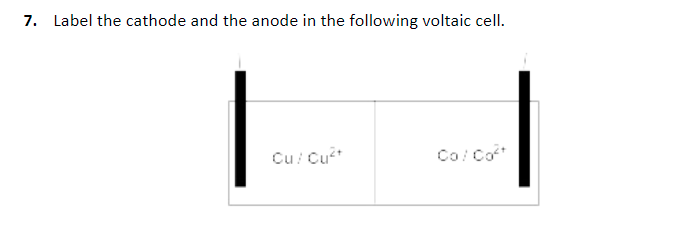
When known, the initial concentrations of ions are usually included in the cell notation, so a more complete cell notation for the cell above is Cu(s) | Cu 2+(aq, 1 M) || Ag +(aq, 1 M) | Ag(s). Note that spectator ions, such as NO 3 −, are not included in the cell notation, and if there are coefficients in a half-reaction, the coefficients are not included (that is, the coefficients of 2 in the silver half-reaction do not appear in the cell notation). The cell notation, Cu(s) | Cu 2+(aq) || Ag +(aq) | Ag(s), is related to the diagram of the voltaic cell. Figure below shows how the cell notation for a voltaic cell relates to various components of the cell. 
Under each beaker, write the appropriate half-rxn. Label the anode and cathode and indicate which species are in the aqueous phase and which are solids (the electrodes). The anode electrode is written to the left, followed by the anode solution, then the salt bridge, then the cathode solution, and, finally, the cathode electrode to the right. The shorthand notation of a galvanic cell specifies the anode on the left, the cathode on the right and the reactants in the half-cell compartments. A galvanic cell has the following composition: Pt(s) I Sn4+ (1M), Sn2+ (1M) II Cu2+ (1M) I Cu(s) From the shorthand notation, draw the galvanic cell. In cell notation, a vertical line, |, denotes a phase boundary and a double line, ||, a salt bridge. Cell notation is an abbreviation that summarizes the important information about a voltaic cell. Drawing a pictorial diagram, like the figure below, to define a voltaic cell takes a lot of time.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
